The Future of Engineered Minichromosomes

Engineered minichromosome is a method of creating small chromosomes that can carry and express multiple genes of interest in plants or animals. These minichromosomes are separate from the normal chromosomes of the host organism and can be used for genetic engineering or synthetic biology purposes.
There are two main ways to produce engineered minichromosomes: the de novo (bottom-up) approach and the top-down approach. The de novo approach involves assembling the essential components of a chromosome, such as centromeres, telomeres, and replication origins, in vitro and then introducing them into a host cell. The top-down approach involves using telomere-mediated chromosomal truncation to cut off the ends of existing chromosomes and attach desired genes to them.
Engineered minichromosomes have several advantages over traditional methods of genetic engineering, such as avoiding the disruption of endogenous genes, allowing the transfer of multiple genes at once, and providing a platform for further modification and manipulation of the minichromosome. They have been successfully generated in several plant species, such as maize, rice, and tobacco, and have potential applications in agriculture, biotechnology, and basic research.
It is a new area of research with the potential to revolutionize genetics, medicine, and agriculture. Traditional gene modification is a slow process. They are small chromosomes that contain a transgene and selectable marker and all of the necessary components required for maintenance in an organism separately from the standard chromosome set. The separation from endogenous chromosomes makes engineered minichromosomes useful in producing transgenic plants.
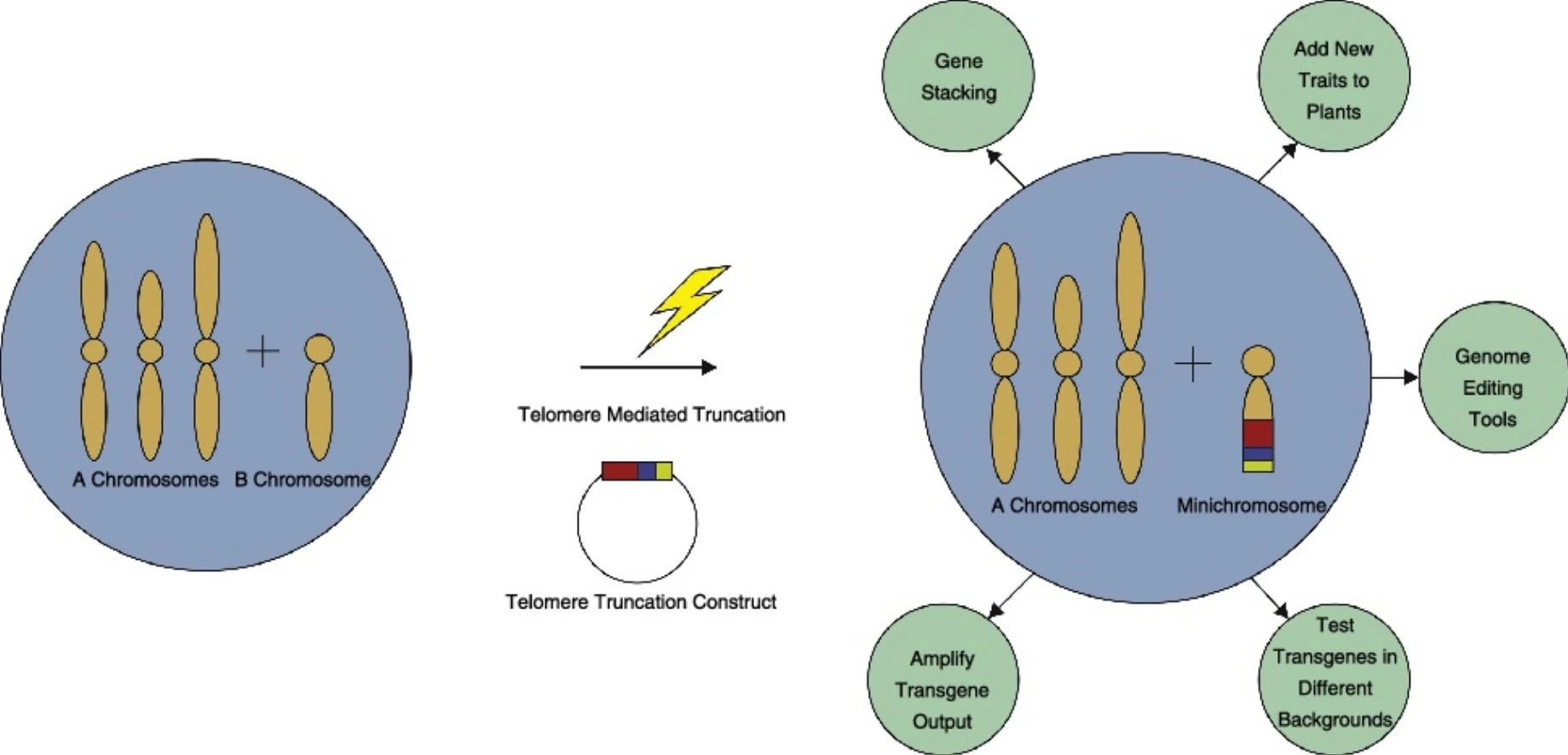
Minichromosomes can add many genes in a much shorter footprint, enabling quick changes. It does not alter plants’ genes in any way. This helps farmers get regulatory approval faster and implement their plans more quickly. These minichromosomes can be swiftly transferred from one unit to another. And increase crops’ nutritional value. They are created by scientists through various techniques, mimicking the structure and function of natural chromosomes but with specific modifications. They feature small structures within cells with little genetic material but can store more information.
It is also known as or related to synthetic chromosomes, minichromids, episomes, telomere-truncated chromosomes, minichromosomes by fragmentation, independent gene expression platforms, stable extrachromosomal elements, mini-me chromosomes, and gene taxis.
By genetically modifying plant genomes, we can:
- Meet growing crop demand
- Reduce diseases
- Enable plantations closer together
- Improve crop’s drought-tolerance
- Improve nitrogen usage
Companies involved in the advancement of engineered minichromosomes:
- Chromatin and Syngenta Biotechnology collaborated to develop minichromosome technology for producing corn, cotton, soybean, and canola.
- Monsanto, Bayer, and BASF are also involved in advancing this technology.
Practical Applications
Some of the practical applications of engineered minichromosomes are:
- They can be used to understand the parameters that allow chromosomes to function. They can be beneficial for studying gene expression and analyzing gene interactions on a large scale.
- They can be used as independent platforms for foreign gene expression without random integration into the normal chromosomes. This can avoid the potential problems of gene silencing, position effects, and pleiotropic effects that may occur with conventional transgenic methods.
- They can combine many useful traits on an independent chromosome that shows no linkage with the remainder of the genome, allowing easy transfer of the transgene set into multiple lines. This can facilitate the development of new varieties with improved agronomic performance, resistance to biotic and abiotic stresses, and enhanced nutritional quality.
- They can be used to add whole biochemical pathways to plants to confer new properties, such as inexpensively producing novel proteins or metabolites in large quantities. This can have applications in biopharmaceuticals, biofuels, and industrial enzymes.
Importance for Our Future
Engineered minichromosome technology is important for our future because it offers a novel way of introducing multiple genes and traits into plants without disrupting their natural genomes. This could have many applications in agriculture, biotechnology, and synthetic biology. For example, engineered minichromosomes could create plants with enhanced resistance to pests, diseases, and environmental stresses or produce valuable proteins or metabolites for human use.
How Does It Work?
The basic steps of these minichromosomes work are as follows:
- A suitable host organism, such as a plant or animal cell, is selected. The host organism should have a well-characterized genome and a reliable transformation method.
- A minichromosome is constructed by either the de novo or the top-down approach. The de novo approach involves assembling the essential components of a chromosome, such as a centromere, telomeres, and replication origins, in vitro and then introducing them into the host cell. The top-down approach involves inserting telomeric sequences into the host genome, which causes the chromosome to break and form a minichromosome with an endogenous centromere.
- It is modified by adding the desired genes and selectable markers using site-specific recombination, transposon insertion, or gene editing techniques. The genes can be arranged in a specific order and orientation and controlled by different promoters and enhancers. The selectable markers can be used to identify and isolate the cells that contain the minichromosome.
- It is transferred to the target organism by direct transformation or crossing with a compatible line. It should be stably inherited and expressed in the offspring and should not interfere with the normal function of the host chromosomes.
Evolve in the Future
Here are the future directions that engineered minichromosomes can take:
- Improve minichromosome production and maintenance efficiency and stability in different plant species. This could involve optimizing the methods of telomere-mediated chromosomal truncation, de novo assembly, or transformation of minichromosomes into plant cells.
- Expand the capacity and functionality of minichromosomes to carry and express multiple genes or complex pathways that confer desirable traits to plants. This could involve using site-specific recombination, gene editing, or synthetic biology techniques to modify the minichromosome contents.
- Develop novel applications of minichromosomes for plant biotechnology, agriculture, and medicine. This could involve using minichromosomes to produce valuable proteins or metabolites, enhance crop yield and quality, confer resistance to biotic or abiotic stresses, or create new varieties of plants with novel characteristics.
Help Organizations/Enterprises
Engineered minichromosomes can help organizations/enterprises in various ways, such as:
- Improve crop performance by introducing multiple genes or pathways that confer desirable traits, such as pest resistance, nutritional enhancement, or yield improvement.
- Produce novel proteins or metabolites in plants that can be used for pharmaceutical, industrial, or environmental purposes.
- Study chromosome structure and function by manipulating and analyzing minichromosomes in different plant species.
- Develop synthetic biology approaches to design and assemble chromosomes to specification.
Driving Adoption
Engineered minichromosomes adoption is driven by several factors, including:
- The need for improving crop quality and production while lowering labor and resource use in agriculture.
- The ability to transfer and express multiple genes of interest in a single entity without disrupting the host genome or affecting its stability.
- Designing and modifying minichromosomes to specification for various purposes, such as conferring new traits, adding whole biochemical pathways, or using plants as factories for specific proteins or metabolites, is possible.
- The possibility to facilitate gene transfer between different plant lines using minichromosomes coupled with haploid induction systems.
- The opportunity to explore the structure and function of chromosomes and centromeres using minichromosomes as a research tool.
Operational Challenges
Some of the challenges in operationalizing engineered minichromosomes are:
- Difficulty in producing stable and functional minichromosomes in different plant species, especially those with large and complex genomes.
- Lack of efficient methods for delivering large DNA fragments containing centromeric and telomeric sequences into plant cells.
- The uncertainty of the minimum size and composition of the centromeric region required for minichromosome formation and maintenance.
- Possibility of interference or competition between the minichromosomes and the endogenous chromosomes for the cellular machinery and resources.
- Potential ethical and environmental concerns associated with using minichromosomes for genetic engineering and biotechnology.
Success Stories
Some of the success stories of engineered minichromosomes in plants are:
In 2007, scientists successfully assembled minichromosomes in vitro using the de novo method. They transformed maize cells with centromeric sequences and screened for plants that assembled autonomous chromosomes de novo. The artificial chromosomes were transmitted mitotically and meiotically.
In 2009, another group of scientists successfully produced minichromosomes in maize using a top-down approach. They used telomere-mediated truncation of chromosomes to create minichromosomes with selectable markers and transgenes. The minichromosomes were stable and heritable in multiple generations.
In 2015, researchers demonstrated the feasibility of using minichromosomes to introduce complex metabolic pathways into plants. They engineered minichromosomes carrying genes for the biosynthesis of artemisinin, a potent antimalarial drug, and transferred them into tobacco plants. The transgenic plants produced artemisinin and its precursors.
In 2018, scientists developed a method to modify minichromosomes in vivo using site-specific recombination. They showed they could add or remove genes or markers from minichromosomes in maize plants without affecting their stability or transmission. This method could enable the rapid and precise manipulation of minichromosomes for various applications.
Types
There are two main types of engineered minichromosomes:
- The top-down approach involves inserting telomeric sequences into the host genome, which causes the chromosome to break and form a minichromosome with an endogenous centromere. This approach is more reliable and plausible in plant systems and can utilize the supernumerary or B chromosomes as starting materials. The limitation of this approach is that it is labor-intensive.
- The de novo approach involves assembling the essential components of a chromosome, such as a centromere, telomeres, and replication origins, in vitro and then introducing them into the host cell. This approach is more difficult than the top-down method due to species incompatibility issues and the heterochromatic nature of centromeric regions.
Both approaches aim to create a chromosomal platform that can carry and express multiple genes of interest in a host organism without disrupting the endogenous genes.
Advantages
Here are some of the advantages of engineered minichromosomes:
- They can transfer and express multiple genes or complex pathways on one engineered chromosome package, conferring desirable traits to plants.
- They do not alter the genes of plants in any manner, resulting in faster regulatory approval and acceptance by farmers.
- They do not have the risk of insertion within a native gene or disrupting endogenous genes, which may negatively affect the host cell.
- They can be used to study the specific chromatin domains inserted into them, such as centromeric regions.
- They can be transferred between lines without the movement of linked genes, which provides a platform for accelerating plant breeding.
- They are independent of the host chromosomes, so they do not interfere with the normal functioning of the plant cells.
- They can carry large amounts of DNA, allowing multiple genes and complex pathways to be inserted.
- They can be modified in vivo, adding or removing genes or markers as needed.
- They can be transferred between different plant species, facilitating the creation of new varieties and hybrids.
Disadvantages
Some of the disadvantages of engineered minichromosomes are:
- The uncertainty of the environmental and health impacts of releasing transgenic plants with engineered minichromosomes into the natural ecosystems.
- The ethical and social implications of creating artificial chromosomes and modifying the genomes of living organisms.
- It is a complex and challenging process that requires high technical skills and resources.
- They may have unpredictable effects on the host genome, such as chromosomal rearrangements, gene silencing, or epigenetic changes.
- They may face ethical, social, and regulatory issues regarding the safety and acceptability of genetically modified organisms.
- They may have limited applicability to some plant species or traits incompatible with minichromosome engineering.
Ethical Concerns
Some of the ethical concerns associated with engineered minichromosomes are:
- The artificial nature: Creating and manipulating chromosomes can be seen as unnatural and violates the integrity of life. There can be questions about the moral status of the organisms that carry the minichromosomes and the potential consequences of altering their genomes.
- The environmental release of GM crops: Minichromosomes could escape from the transgenic plants and transfer to other species, causing ecological disruption and gene flow. There may also be fear that the minichromosomes could confer undesirable traits on the plants, such as invasiveness, allergenicity, or toxicity.
- The negative impact on human health: There may be doubt about the safety and efficacy of the products derived from minichromosome-bearing plants, such as biopharmaceuticals, biofuels, and industrial enzymes. The possible health risks of consuming or being exposed to GM crops include allergic reactions, gene transfer, or antibiotic resistance.
- The negative impact on farmers: Concerns about the social and economic implications of the minichromosomes for farmers, especially in developing countries. Also, it could increase the farmers’ dependency on the corporate firms that own the patents and licenses and reduce local crops’ and cultures’ diversity and sovereignty.
- The excessive domination of the corporate firms: There can be opposition to the monopoly and control of minichromosomes by multinational agribusiness corporations. Some may accuse the corporations of exploiting the farmers, consumers, and regulators for their profits and interests and challenge the corporations’ transparency and accountability regarding research, development, and regulation.
- The religious and cultural concerns: Some people may have religious or cultural objections to the minichromosomes based on their beliefs and values. They may consider the technology violating creation’s divine or natural order. They may also reject the technology as incompatible with their traditions and customs.
Governance and Regulation
Here are the possible aspects to govern and regulate engineered minichromosomes:
- The ethical, social, and environmental implications of creating and using artificial chromosomes in plants. For example, how would this affect the biodiversity, biosafety, and ownership of plant genetic resources? What are the potential risks and benefits for human health, food security, and environmental sustainability? How would this impact the cultural and religious values of different communities?
- The legal and policy frameworks govern engineered minichromosome development, approval, and commercialization. For example, what laws and regulations apply to this in different countries and regions? How would this comply with the international agreements and conventions on plant genetic engineering, such as the Cartagena Protocol on Biosafety and the Nagoya Protocol on Access and Benefit Sharing? What are the gaps and challenges in the current legal and policy systems need to be addressed?
- The scientific and technical standards and guidelines ensure quality, safety, and efficacy. For example, what are the best practices and methods for producing, testing, and using engineered minichromosomes in plants? How would this be monitored and evaluated for its performance and impact? What are the criteria and indicators for assessing the safety and effectiveness of engineered minichromosomes in plants?
However, this issue has no one-size-fits-all solution, as different contexts and situations may require different approaches and strategies. Therefore, it is important to engage in a transparent, inclusive, and participatory dialogue among all the relevant stakeholders, such as scientists, policymakers, regulators, industry, farmers, consumers, civil society, and the public, to ensure that this technology is developed and used in a responsible, ethical, and sustainable manner.
History
Engineered minichromosomes are a relatively new and emerging field in plant biotechnology and synthetic biology. The history of this technology can be traced back to the following milestones:
In 1983, Murray and Szostak reported the first generation of artificial chromosomes in yeast by assembling the essential components of a chromosome (centromere, telomere, origin of replication, and selectable marker) in vitro and introducing them into yeast cells.
In 1992, Farr et al. discovered that transforming telomere sequences into human cells could induce chromosomal truncation at integration sites.
In 1997, Harrington et al. produced artificial chromosomes in human cells by transforming centromere sequences and allowing the cells to assemble them into functional chromosomes.
In 1998, Preuss et al. demonstrated the feasibility of generating minichromosomes in plants using telomere-mediated chromosomal truncation in Arabidopsis thaliana.
In 2007, Gaeta et al. reported maize’s first successful production of engineered minichromosomes by transforming telomere sequences linked to transgenes and an endogenous centromere.
In 2012, Yu et al. showed that engineered minichromosomes in maize could be modified in vivo by site-specific recombination and selectable marker removal.
In 2015, Birchler reviewed the current status and prospects of engineered minichromosome technology in plants.
Tools and Services
Engineered minichromosome tools and services are products or methods that can help create, manipulate, or use engineered minichromosomes in plants or animals. Example:
- Telomere-mediated chromosomal truncation: This technique uses telomere sequences to cut off the ends of existing chromosomes and attach desired genes to them. This can generate minichromosomes with an endogenous centromere and a selectable marker.
- Site-specific recombination systems: These are systems that use enzymes such as Cre or FLP to insert or remove transgenes from minichromosomes in vivo. This can increase the utility and flexibility, allowing further modification and manipulation.
- Binary bacterial artificial chromosome vectors: These vectors can carry large DNA fragments and simultaneously introduce multiple transgenes into minichromosomes. These vectors can also be used to clone and manipulate minichromosomes in bacteria.
- Haploid induction systems: These systems can produce haploid plants from diploid plants using various methods such as pollination with irradiated pollen, anther culture, or microspore culture. These systems can facilitate the transfer of minichromosomes between different plant lines and increase the efficiency of breeding.
How to Get Started?
To get started with engineered minichromosome technology, you must have some basic knowledge and skills in molecular biology, genetics, and biotechnology. You will also need access to a suitable host organism, such as a plant or an animal cell, and the necessary tools and materials to construct and modify minichromosomes. Here are some possible steps to follow:
- Learn about the structure and function of chromosomes, especially the centromere, telomere, and replication origin regions. You should also understand how chromosomes are inherited and expressed in the host organism.
- Decide which approach you want to use to create minichromosomes: the de novo or the top-down method. The de novo method involves assembling the essential components of a chromosome in vitro and then introducing them into the host cell. The top-down method consists of inserting telomeric sequences into the host genome, which causes the chromosome to break and form a minichromosome with an endogenous centromere.
- Design and synthesize the desired genes and selectable markers to add to the minichromosome. You should also consider the order and orientation of the genes and the promoters and enhancers that control their expression. You can use site-specific recombination, transposon insertion, or gene editing techniques to insert the genes and markers into the minichromosome.
- Transform the minichromosome into the target organism by direct transformation or crossing with a compatible line. You should then select and isolate the cells that contain the minichromosome and verify its structure and function. You can use molecular and cytological techniques, such as PCR, Southern blot, FISH, and karyotyping, to analyze the minichromosome. You can also evaluate the phenotypic effects of the minichromosome, such as the expression of the transgenes and the traits they confer.

Best Practices
Some of the best practices for getting the most from engineered minichromosomes are:
- Choose the appropriate method for creating minichromosomes depends on the plant species and the desired traits. For example, telomere-mediated truncation is more reliable and efficient than de novo assembly in most plant systems.
- Select suitable centromere sequences that support stable and functional minichromosomes in different plant species. For example, using native centromeres from the host plant or closely related species or synthetic centromeres that can mimic the essential features of natural centromeres.
- Optimize the size and content of the minichromosomes to avoid unnecessary DNA or potential interference with the host genome. For example, it uses minimal centromeres, removes non-essential sequences, and avoids repeated or transposable elements that could cause instability or recombination.
- Develop efficient methods for transferring minichromosomes between different plant species, either by sexual or asexual means. For example, using pollen or ovule culture, protoplast fusion, or biolistic transformation.
- Implement site-specific recombination systems that allow adding or removing genes or markers from minichromosomes in vivo. For example, using Cre/loxP, Flp/FRT, or PhiC31/attP systems.
- Evaluate the performance and safety of the transgenic plants with minichromosomes under different environmental and agronomic conditions. For example, testing the stability and inheritance of the minichromosomes, the expression and regulation of the transgenes, and the potential impacts on the host genome, the plant phenotype, and the ecosystem.
- Follow the ethical and legal guidelines for using and releasing transgenic plants with minichromosomes. For example, obtaining the necessary approvals and permits, ensuring the traceability and labeling of the products, and engaging with the public and stakeholders.
Related Terms
Some of the terms related to engineered minichromosomes are:
- Minichromosome: A small chromatin-like structure resembling a chromosome consisting of centromeres, telomeres, and replication origins but little additional genetic material.
- Centromere: A region of DNA that links sister chromatids and attaches them to the spindle fibers during cell division.
- Telomere: A repetitive DNA sequence at the end of a chromosome that protects it from degradation and fusion.
- Replication origin: A specific sequence of DNA where DNA replication begins.
- Transgene: A gene transferred from one organism to another, usually by genetic engineering.
- Selectable marker: A gene that confers a trait that allows the selection of cells that carry it, such as antibiotic resistance or herbicide tolerance.
- De novo approach: A method of producing minichromosomes by assembling the minimum constituent parts of a chromosome in vitro and then introducing them into a host cell.
- Top-down approach: A method of producing minichromosomes using telomere-mediated chromosomal truncation that cleaves chromosome arms and simultaneously places desired transgenes linked to an endogenous centromere.
- Telomere-mediated chromosomal truncation (TMCT) generates truncation by selectively transforming telomeric sequences into a host genome.
- Site-specific recombination: A type of genetic recombination that occurs at specific DNA sequences and is mediated by enzymes called recombinases.
- Synthetic biology: A field of science that involves designing and creating new biological systems or components that do not exist in nature.
Learn more
Research Papers
- Engineered minichromosomes in plants by James A. Birchler and Nathan C. Swyers: Summarizes the approaches and advantages of engineered minichromosomes in plants and the behavior and potential applications in basic and applied research.
- Construction and behavior of engineered minichromosomes in maize by James A Birchler, Weichang Yu, Fangpu Han, Zhi Gao, and Juan M Vega: Reports using telomere-mediated truncation to produce minichromosome platforms in maize by cleaving both normal and supernumerary chromosomes and introducing site-specific recombination cassettes for future manipulations.
- Engineered plant minichromosomes by Andreas Houben, Michael Florian Mette, Chee How Teo, Inna Lermontova, and Ingo Schubert: Describes the construction of maize minichromosomes using shuttle vectors that contain native centromeric segments, replication origins, selectable marker genes, and telomeric repeats. Microprojectile bombardment introduced These vectors into the scutellar cells of immature maize embryos.
- Engineered Minichromosomes in Plants: Structure, Function, and Applications by Nathaniel D Graham, Jon P Cody, Nathan C Swyers, Morgan E McCaw, Changzeng Zhao, James A Birchler: Reviews the current state of the art of engineered minichromosomes in plants, their structure, function, and applications. It also describes techniques and tools for creating and manipulating minichromosomes, such as site-specific recombination systems and binary bacterial artificial chromosome vectors.
- Engineered Plant Minichromosomes: A Bottom-Up Success? by Andreas Houben, R. Kelly Dawe, Jiming Jiang, and Ingo Schubert: Evaluates the progress and potential of engineered plant minichromosomes, their advantages and disadvantages, and their prospects. It also compares the bottom-up and top-down approaches for creating minichromosomes and suggests some directions for further research.
- Engineered Plant Minichromosomes: A Resurrection of B Chromosomes? by Andreas Houben and Ingo Schubert: Reviews the current status and prospects of engineered minichromosomes in plants, comparing them with the naturally occurring supernumerary B chromosomes. The article also discusses the mechanisms and factors involved in forming and maintaining engineered minichromosomes and their potential applications in biotechnology and basic research.
Books
- Recent Advancements in Gene Expression and Enabling Technologies in Crop Plants: Overviews of engineered minichromosomes’ potential applications and challenges in plant biotechnology and synthetic biology.
- Plant Chromosome Engineering: Methods and Protocols: Describes the methods and techniques for generating and modifying engineered minichromosomes in maize using telomere-mediated truncation and site-specific recombination.
Articles
- Minichromosomal Technology in Agriculture – fsii: Explains the basics of minichromosomes, their applications in agriculture, and their advantages over traditional genetic engineering methods. It also gives some examples of crops that have been improved using minichromosomes, such as Arctic apples that do not brown.
- MINICHROMOSOME TECHNOLOGY – AGRIALLIS: Provides a detailed overview of minichromosome technology, its history, its methods, its benefits, and its challenges. It also discusses some ethical and social issues related to this technology.
- Editorial: Recent advances in plant genetic engineering and synthetic biology: Introduces a collection of papers showcasing recent developments and innovations in plant genetic engineering and synthetic biology, including engineered minichromosomes, genome/gene editing, protein-domain specific technology, and virus-induced silencing. The article also highlights the challenges and opportunities for these technologies in improving crop performance and quality.
I hope you enjoyed this insightful discourse. I believe that open dialogue and diverse perspectives are essential for fostering a deeper understanding of complex issues. Therefore, encourage you to share your thoughts and opinions in the comments below. Engage with other readers, challenge assumptions, and contribute to a rich and meaningful discussion. Your insights are valuable, and welcome you to participate actively in this intellectual exchange.
Together, we can explore new ideas, expand our horizons, and generate fresh perspectives.

Learner-in-Chief at Future Disruptor. A futurist, entrepreneur, and management consultant, who is passionate about learning, researching, experimenting, and building solutions through ideas and technologies that will shape our future.
Subscribe to the Future Disruptor newsletter.






Leave a Reply